DCM & SyMMES
- Share
- Share on Facebook
- Share on LinkedIn
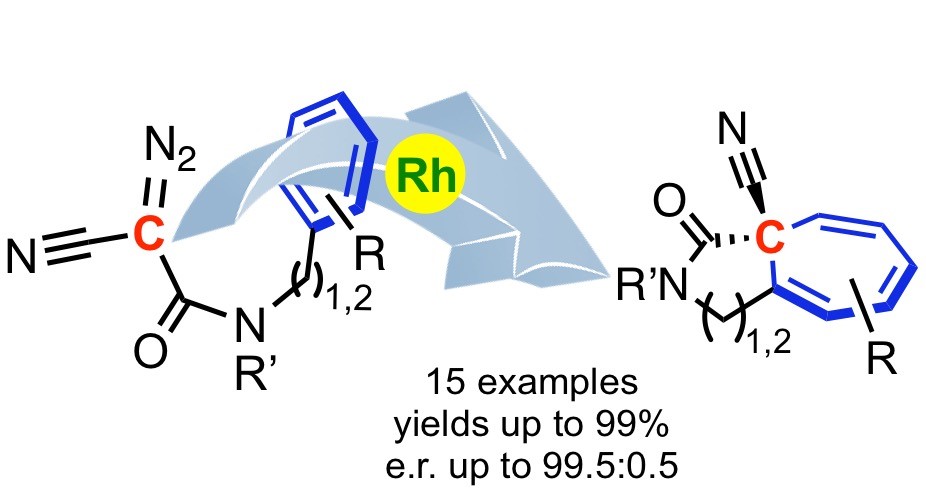
Bicyclic compounds bearing a quaternary stereogenic center have been obtained using asymmetric intramolecular Buchner reaction with excellent yields and level of enantioselectivity. X-ray crystallography determination of the absolute configuration of one product has led to the serendipitous observation of an unusual behavior within the crystal structure, with equilibrating norcaradiene and cycloheptatriene valence isomers at the solid state, as well as an even more unexpected intermediate form. DFT calculations were performed to support these observations.
Asymmetric Intramolecular Buchner Reaction: From High Stereoselectivity to Coexistence of Norcaradiene, Cycloheptatriene, and an Intermediate Form in the Solid State, Dr. B. Darses, Dr. P. Maldivi, Dr. CC Philouze, Dr. P. Dauban, Prof. J.-F. Poisson, Org. Lett. 2021, 23, 300-304.
- Share
- Share on Facebook
- Share on LinkedIn