- Share
- Share on Facebook
- Share on LinkedIn

Extracellular Vesicles (EVs) are nanoscale lipid particles secreted by cells and actively participating to intercellular communication. They have received increasing attention over the past decade as non-invasive biomarkers easily available from liquid biopsy, and they are considered for diagnostic applications. Isolation of such biomarkers however remains challenging mainly due to EVs heterogeneity, especially in size, and biofluids complexity. New methods are required to perform simple, efficient and reproducible EVs isolation. In this PhD thesis, a coupled approach is proposed, based on an aptamer-supported capture of EVs, combined with a Deterministic Lateral Displacement (DLD) size sorting system, in order to demonstrate EVs fractionation according to their protein expression.
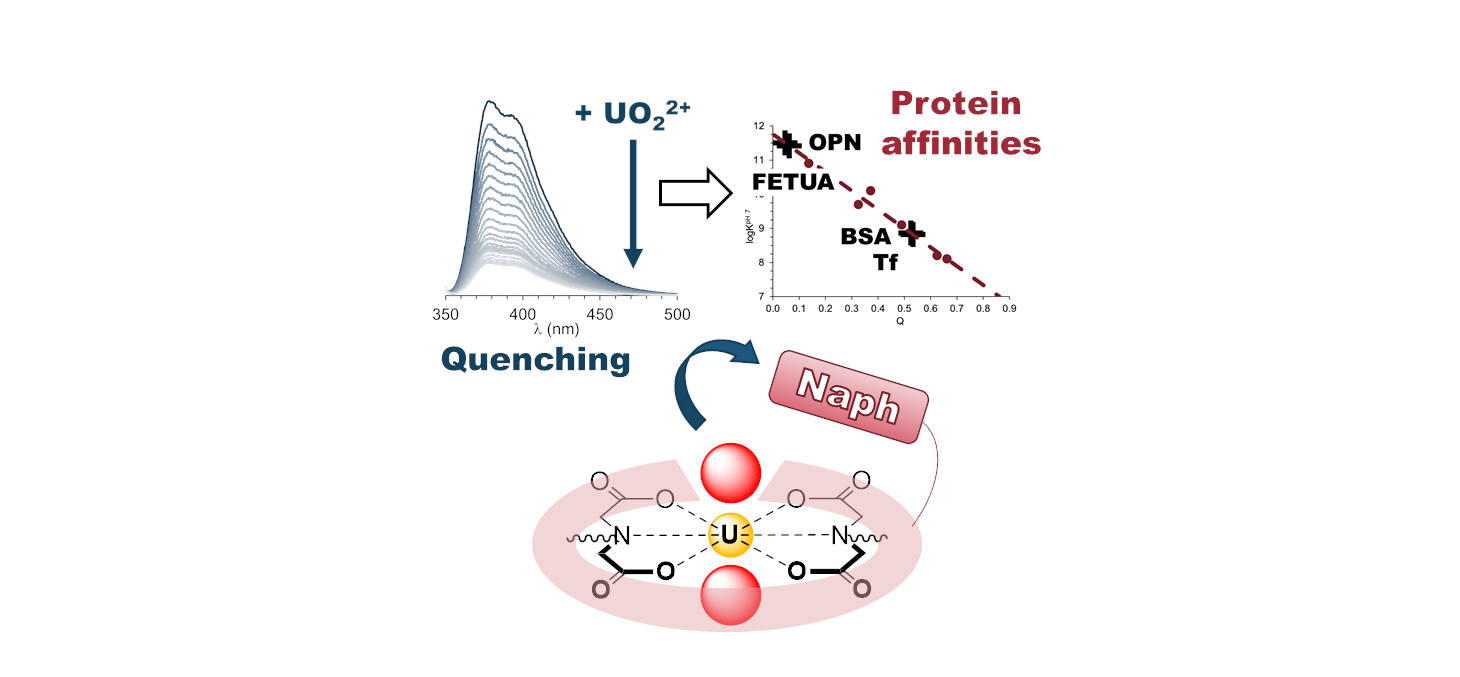
Uranium is naturally present in the environment as the dioxo cation uranyl. Understanding its interaction with proteins is key to decipher the mechanisms underlying its toxicity and to develop decorporation strategies. In this process, determining affinities of proteins for uranyl is important, but requires specific equipment and expertise. In this work, we developed a simple fluorescence-based assay to determine uranyl-protein affinities in solution, in physiological conditions, and at equilibrium.
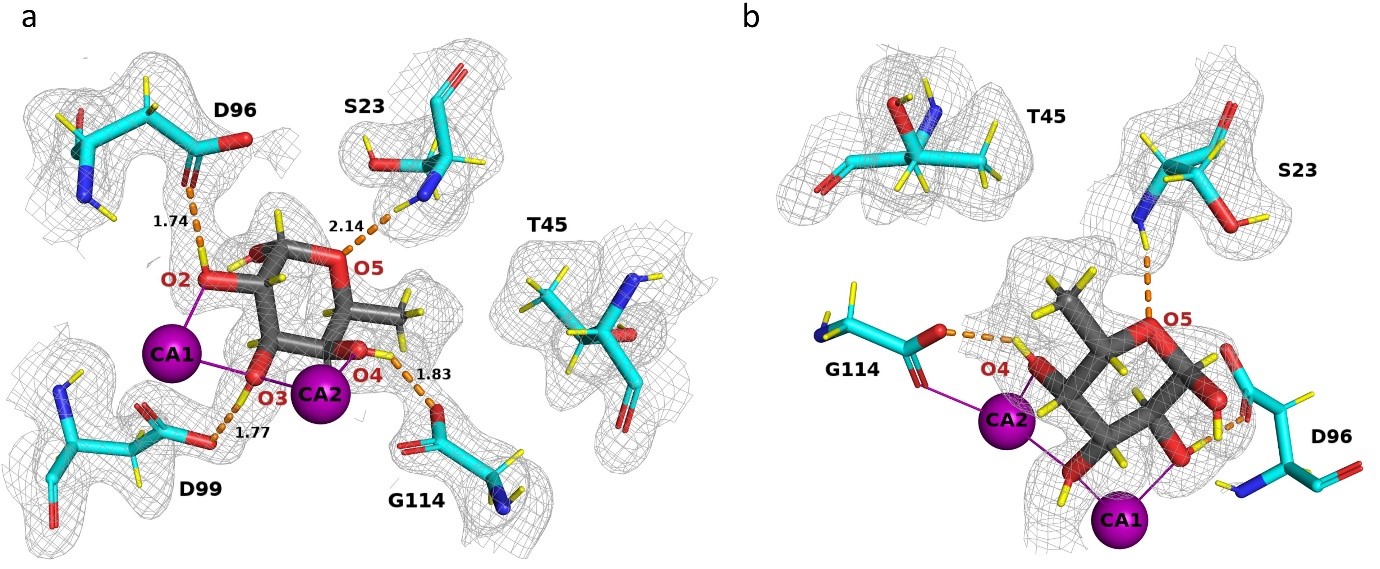
In view of the threat to human health caused by bacterial antibiotic resistance, new strategy, such as anti-infectious compounds, are of high interest. Pseudomonas aeruginosa, a major cause of nosocomial infections, uses carbohydrate-binding proteins (lectins) as part of its binding to host cells. A soluble fucose-binding lectin, LecB, displays a unique carbohydrate-binding site that involves two closely located calcium ions bridging between the ligand and protein, providing specificity and unusually high affinity. Several high resolution X-ray crystal structures of LecB/fucose complex are available. However, the location of hydrogen atoms, that are crucial in the interaction mechanisms are not visible by X-ray crystallography.
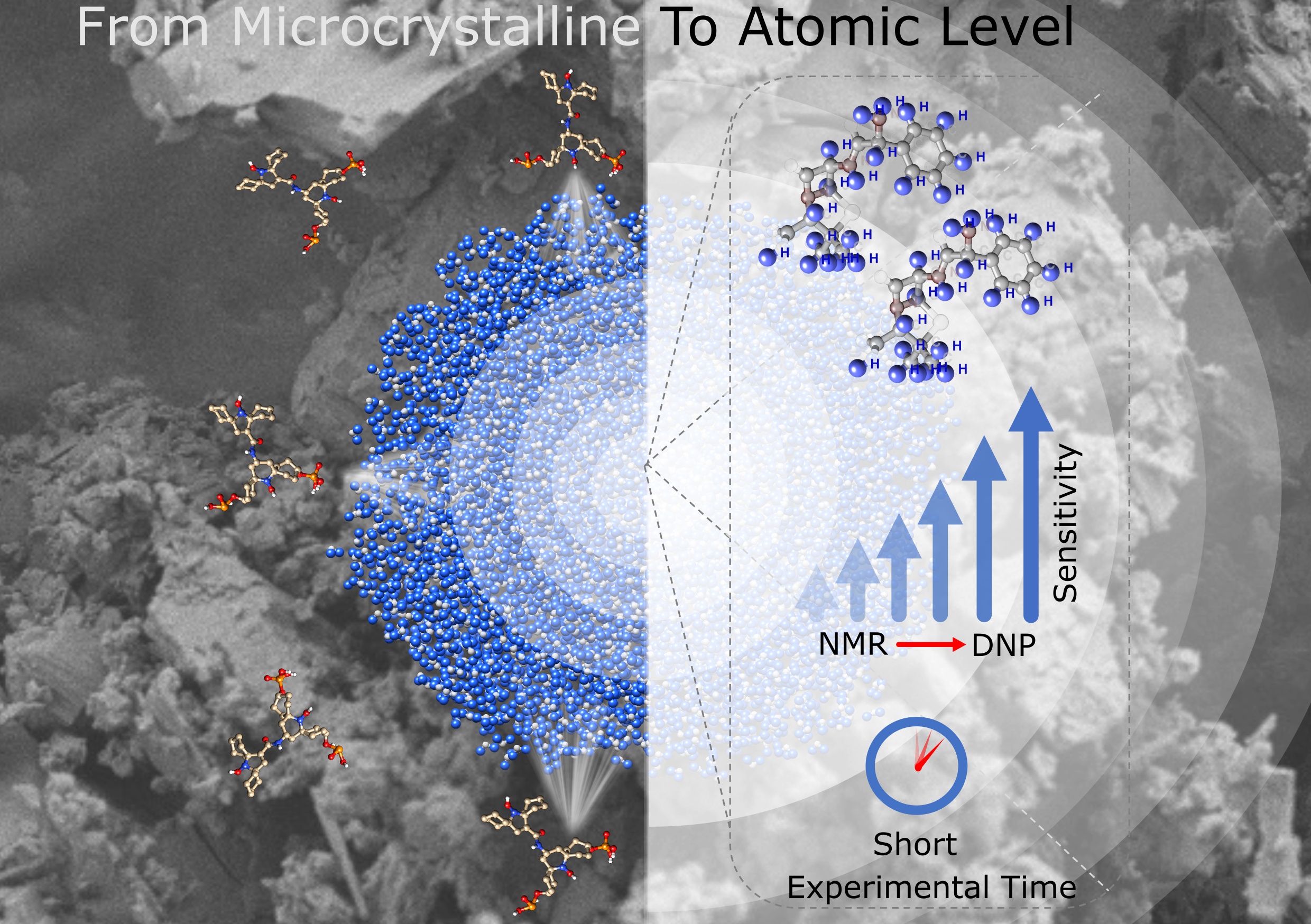
Efficiently hyperpolarizing proton-dense molecular solids through dynamic nuclear polarization solid-state NMR is still an unmet challenge. Polarizing agents (PAs) developed so far do not perform well on proton-rich systems, such as organic microcrystals and biomolecular assemblies. In this paper, we introduce a new PA, cAsymPol-POK, and report outstanding hyperpolarization efficiency on a protein and pharmaceutical drugs at high magnetic fields and fast magic angle spinning frequencies.
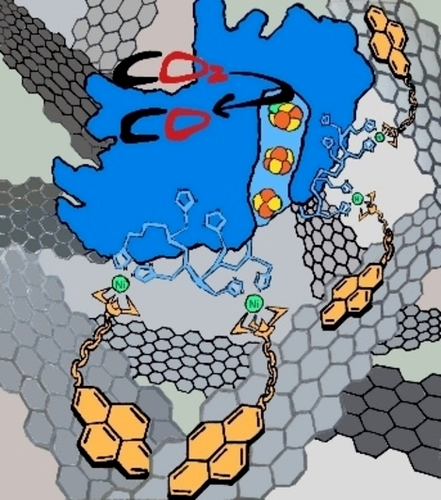
An original 1-acetato-4-(1-pyrenyl)-1,4,7-tria- zacyclononane (AcPyTACN) was synthesized for the immobilization of a His-tagged recombinant CODH from Rhodospirillum rubrum (RrCODH) on carbon-nanotube electrodes. The strong binding of the enzyme at the Ni-AcPyTACN complex affords a high current density of 4.9 mA cm-2 towards electroenzymatic CO2 reduction and a high stability of more than 6 × 106 TON when integrated on a gas-diffusion bioelectrode.
Solid-state NMR enables the investigation of biological systems within their native context, e.g. lipid membranes, viral capsid assemblies, and cells. However, such investigations often suffer from low sensitivity due to low concentrations. Using dynamic nuclear polarization (DNP), the experimental time can be reduced by orders of magnitude, broadening the application scope. In this review, we outline strategies and present an overview of investigations where DNP has led to unique insights.
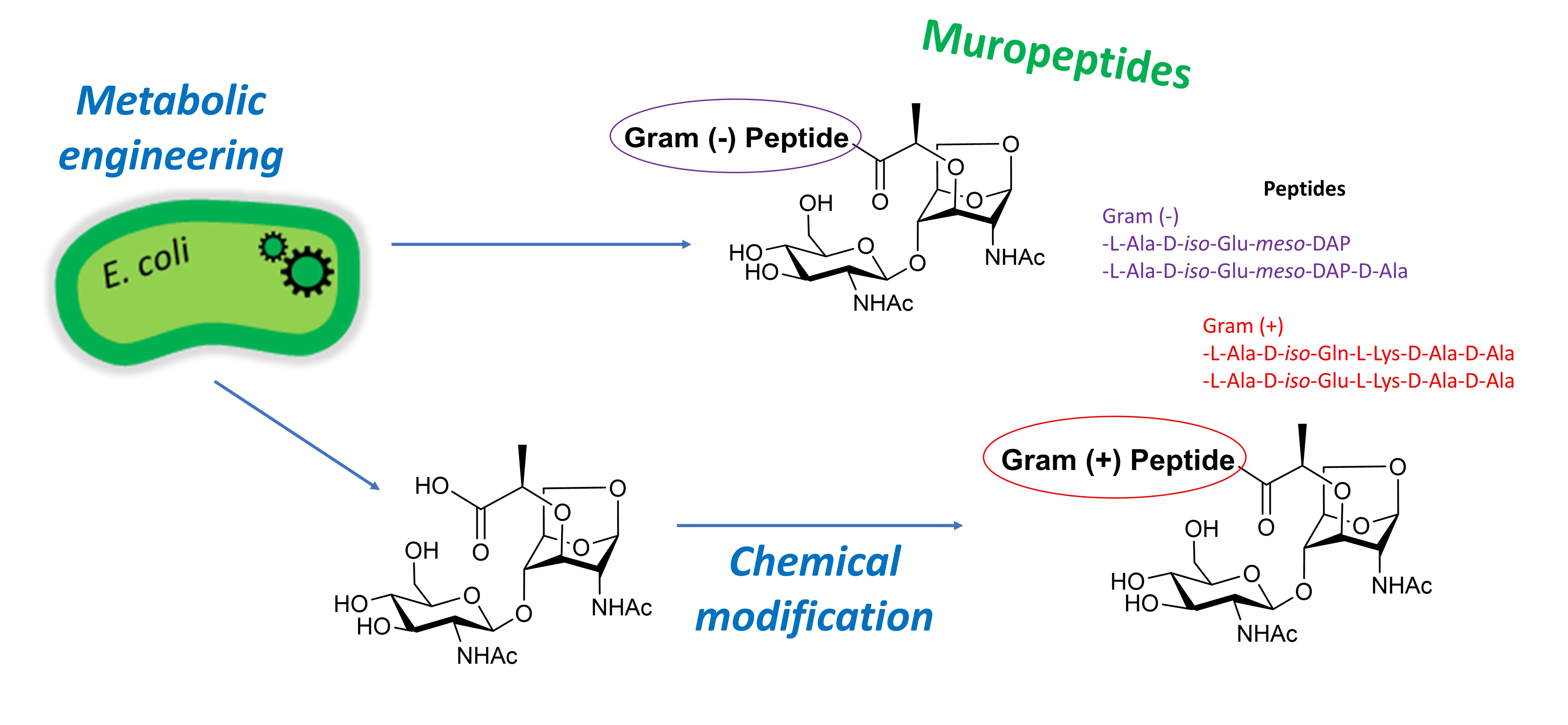
Muropeptides are short peptidoglycan fragments released from the cell wall of bacteria as part of their metabolism or as a result of biological stresses. These compounds trigger immune responses in mammals and plants. In bacteria, they play a major role in the induction of antibiotic resistance. In order to better understand these processes, we developed a new synthetic approach which combines synthetic biology and glycochemistry to prepare Gram (–) and Gram (+) muropeptides.
- Share
- Share on Facebook
- Share on LinkedIn